What makes radium unique?

- Characteristics
- Isotopes
Unusual Characteristics:
- Radium is luminescent, meaning it glows in the dark giving off a faint blue/green color.
- One million times more reactive than uranium.
Crystal Shape:
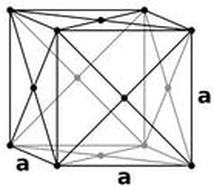
Radium's crystal structure is cubic.
Isotopes:
- Radium has about 33 known isotopes.
- All of them are very radioactive.
- Four of them are found in nature: radium-223, radium-224, radium-226, and radium-228.
- Most common and most stable= radium-226, discovered by the Curies. Used to make radon gas.
